Neuromodulation & Pelvic Health

InterStim X™
Meet InterStim X™ our newest recharge-free system for patients living with bladder and bowel control symptoms. InterStim X™ is the next generation of the first and most established Sacral Neuromodulation technology.


Medtronic Intellis SCS
Chronic Pain Management: IntellisTM is the world's smallest implantable neurostimulator, offering 95% capacity at 9 years1, personalised programming, and unmatched MRI access.

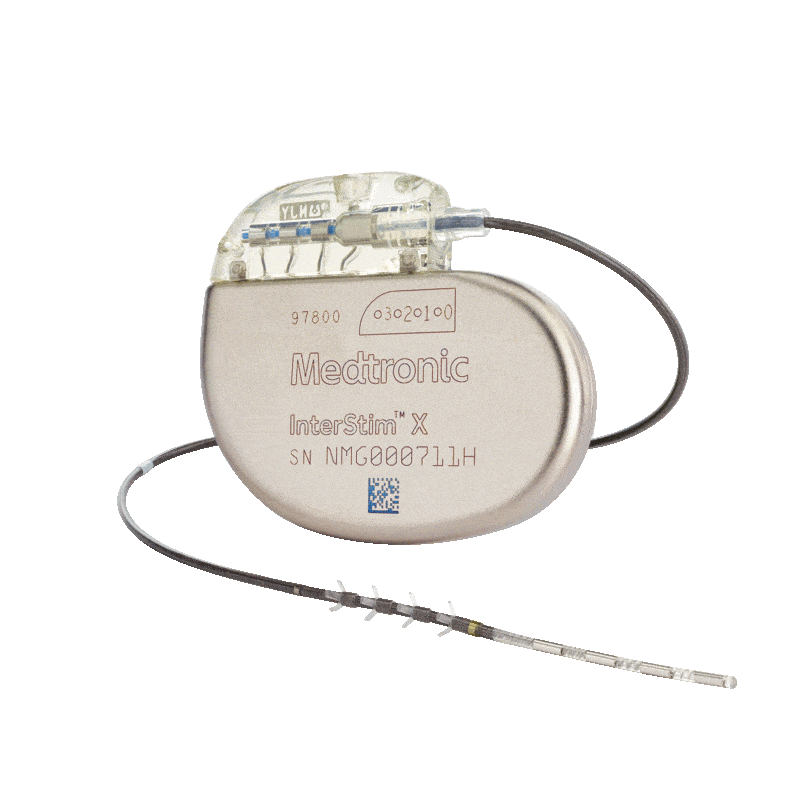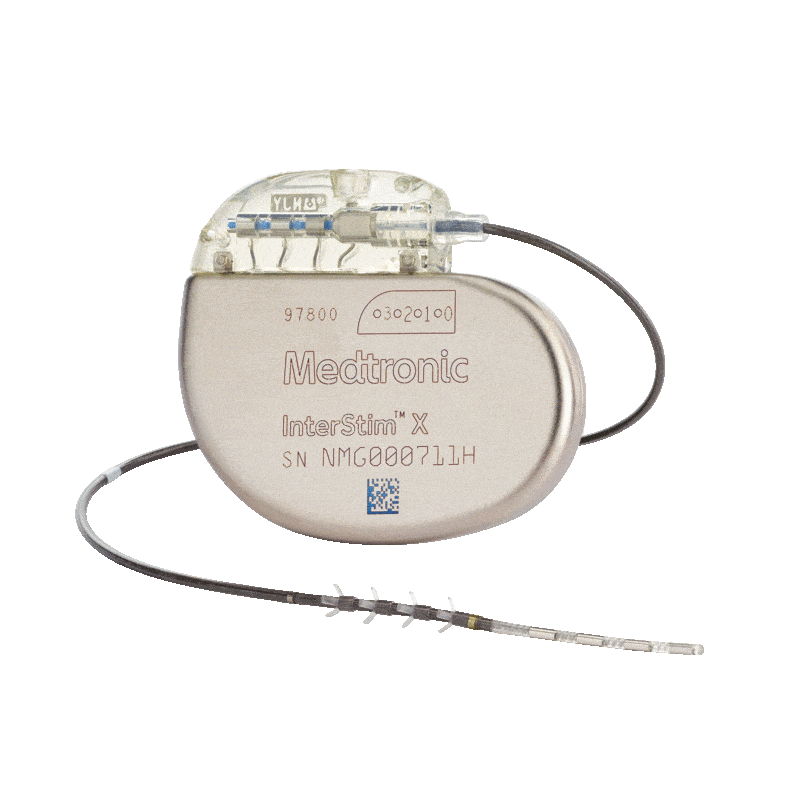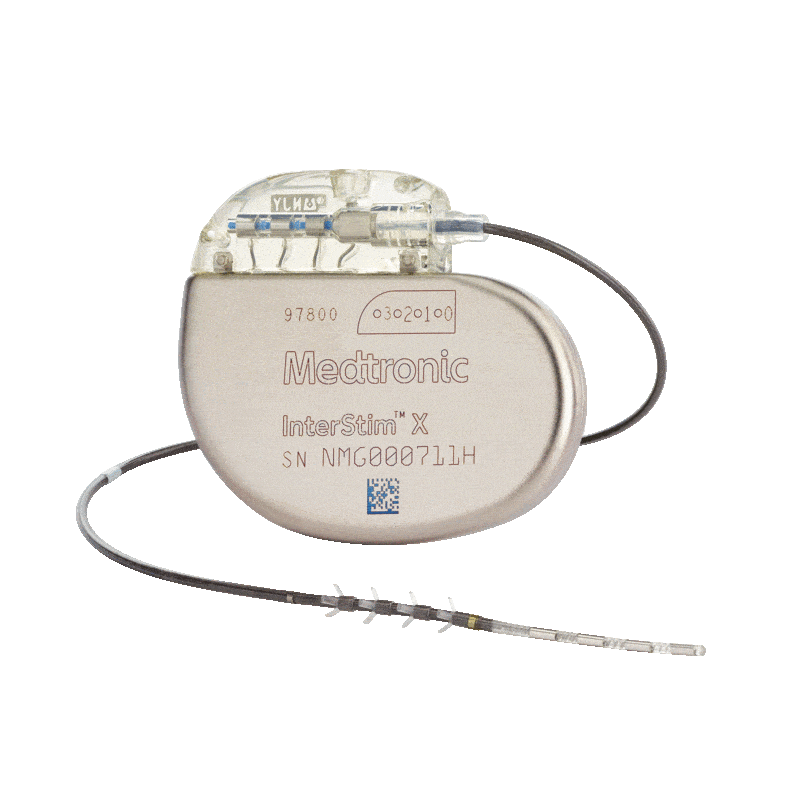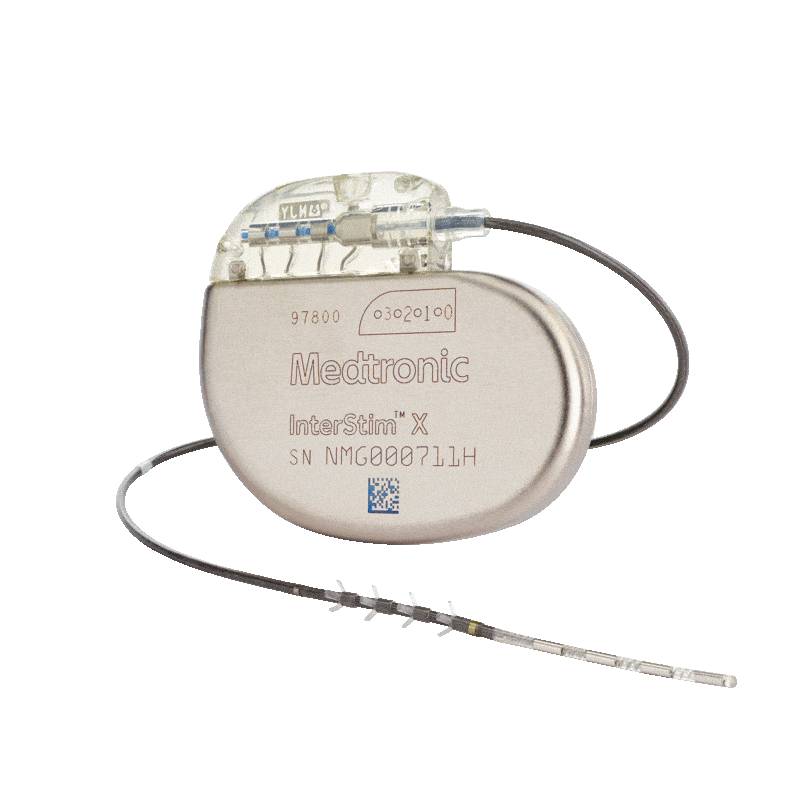
Medtronic Intestim II
Incontinence Management: The implanted InterStim™ II system electrically stimulates the sacral nerve, which is thought to normalise neural communication between the bladder and brain and between the bowel and brain.

Medtronic SynchroMed II
Severe Spasticity Management: The SynchroMed™ II programmable, implantable pump delivers drug to the intrathecal space via an implanted catheter. SynchroMed™ II pumps can be managed using the Control Workflow™ approach, which is designed to help eliminate systemic opioids and provide effective pain relief.

Percept™ PC Neurostimulator with BrainSenseTM
Parkinson's Disease and Movement Disorders Management: Personalize therapy with confidence. The first complete DBS system with sensing, directionality, and visual programming gives you access to the data-driven insights you need to make informed decisions.

Percept™ RC
Neurostimulator with BrainSenseTM
The Percept™ RC neurostimulator with exclusive BrainSense™ technology† is the first complete rechargeable DBS system with sensing, directionality, and advanced programming capabilities. The device gathers real-time data to offer insights into the patient’s condition, inside and outside the clinic, allowing you to adapt therapy to your patients’ evolving needs over time.

URIS Technology
The URIS I nerve stimulator is intended for the treatment of lower urinary tract symptoms (urgency, frequency, nocturia, incontinence) using non-invasive electrical transcutaneous neuromodulation of the peripheral nerves or through percutaneous electrical stimulation of the tibial nerve (the treatment method is referred to as percutaneous tibial nerve stimulation (PTNS)), which serves to influence the nerve control of the lower urinary tract at the peripheral, spinal and supraspinal levels (insula, operculum , cingulum, supplementary motor area, middle frontal gyrus).
The URIS® device operates on a principle of electrical transcutaneous nerve modulation (eTNM®). Through specially designed electrodes, that do not compromise the integrity of the skin, URIS® can selectively stimulate nerve pathways to the brain. Such precisely targeted stimulation activates brain centers that are in decline, or to inhibit overly active brain centers in order to restore balance to the brain.
The URIS® device is the first clinically tested neuromodulation device that uses a closed-loop biofeedback system and is designed to allow treatment from the comfort of one’s home. Patients are provided with a treatment tailored to their conditions without the need to travel to a doctor.


Building Lasting Relationships
Reach out to us today to discover how our medical devices can enhance your healthcare operations. We are eager to assist you in advancing patient care and achieving your organizational goals.


